
Abdominal Aortic Calcification (AAC): The “Rust Line” That Can Quietly Change Spine Surgery Risk
A city can look perfectly functional from street level: traffic lights working, sidewalks intact, buildings standing. But civil engineers worry about something else: the water main beneath it all. If that main is stiff, narrowed, and lined with mineral scale, the city doesn’t fail in one dramatic moment. Instead, everything becomes less resilient: pressure drops under demand, repairs take longer, and downstream neighborhoods become more vulnerable when stress hits.
That’s a useful way to think about the abdominal aorta in spine surgery. The spine (bones, discs) may be what we “see” and operate on, but the vascular supply is part of the hidden infrastructure that determines how well tissues tolerate surgical stress, heal, and remodel.

What the aorta is and when are we talking about aortic abdominal calcification?
The aorta is the biggest blood vessel in your body. Think of it like a huge highway pipe that carries blood from your heart down into your belly(abdomen) and to your legs.
Abdominal aortic calcification isa fancy way of saying: there are hard, chalky “stone-like” bits stuck to the inside wall of this big blood tube.
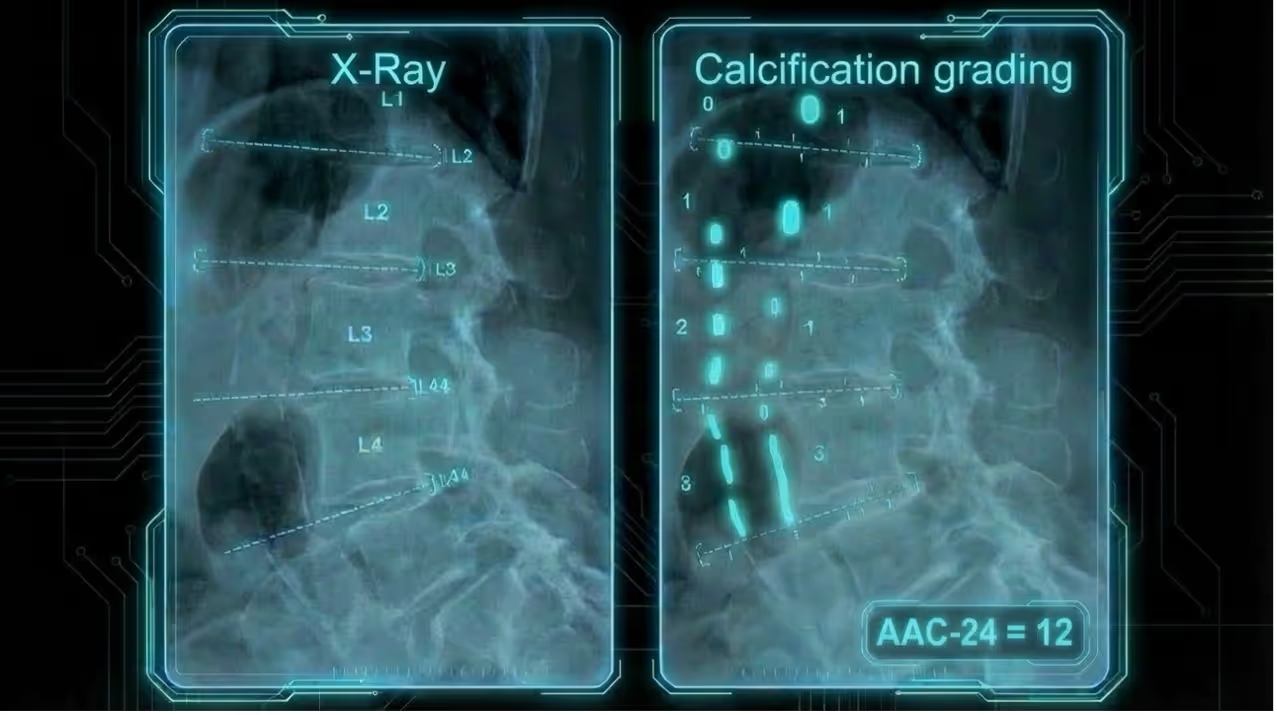
So, what is the buildup made of? It’s called plaque, a mix of fats and other stuff. Overtime, calcium can stick to it and make it hard, like mineral crust inside an old water pipe. On a side-view (lateral) X-ray of the lower back, doctors can see this hard buildup because it shows up bright white.
It can look like:
- a bright, curving pair of white lines, like train tracks (“railroad tracks”), or
- little broken white pieces, like segments or patches in front of the spine bones [1]
Abdominal aortic calcification matters because it’s a clue that blood vessels in the whole body are getting stiff and “gunky” with age. That can mean blood might not flow as smoothly, which can make it harder for the body to handle surgery and heal afterward[1].
How to measure abdominal aortic calcification?
Abdominal aortic calcification can be checked using a side-view X‑ray of your lower back (a lateral lumbar X‑ray): they look for little white, chalky lines in the big blood tube (the abdominal aorta) that runs in front of the lumbar spine, then they give it a score overall (a number from 0 to 24, where 0 = no chalk and **24 = lots of chalk) so they can describe it the same way every time [1–3].
Why abdominal aortic calcification is a high-leverage risk signal surgeons can use
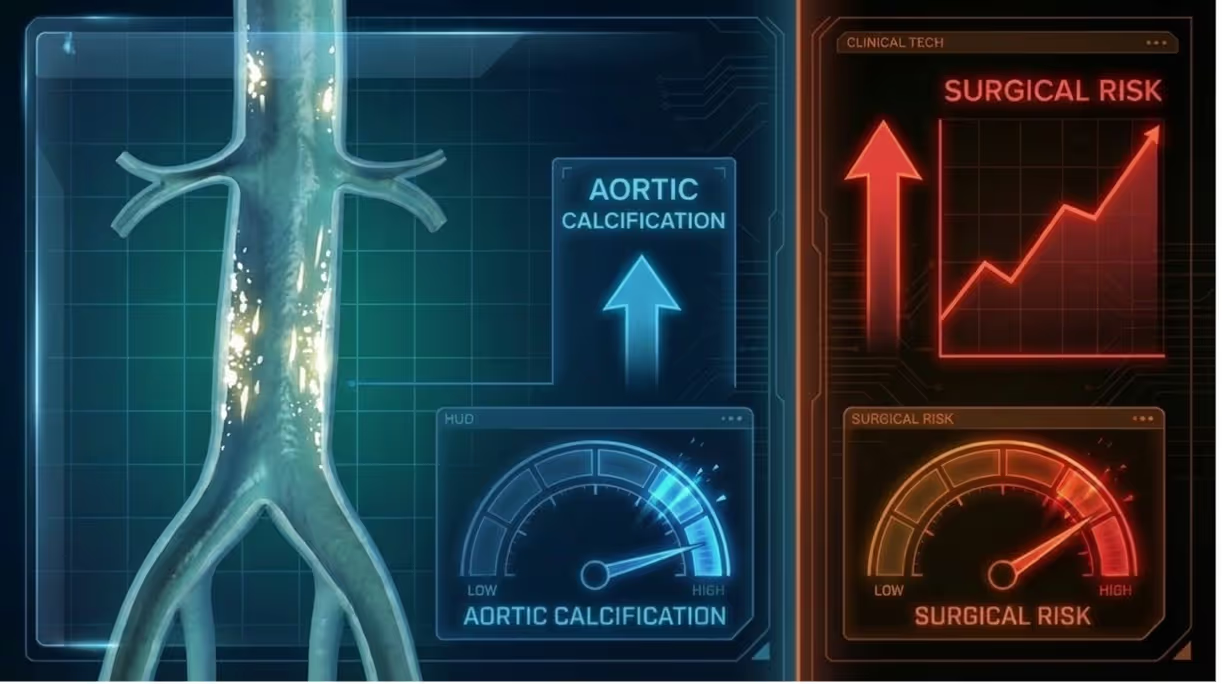
Like paraspinal muscles, abdominal aortic calcification is the vascular version of hidden biological cues that shape surgical outcomes. Abdominal aortic calcification is a visible marker of systemic health that can alter surgical risk even when the spine problem looks straightforward.
With that in mind, here are specific findings from scientific papers that connect abdominal aortic calcification to surgical outcomes:
- Abdominal aortic calcification can act like a practical “vascular red flag” in spine patients [2]
- More abdominal aortic calcification → higher risk the fusion won’t heal fully (pseudarthrosis). In posterior lumbar fusion, higher abdominal aortic calcification severity independently predicted impaired fusion, especially posterolateral fusion (odds ratios reported around 1.10–1.16 per score step, depending on fusion type/outcome) [2]
- More abdominal aortic calcification → more blood loss during posterior spinal fusion. Abdominal aortic calcification independently predicted greater peri‑op blood loss (about +4.46% estimated blood loss/total blood volume after adjustment) [3]
- Higher abdominal aortic calcification (atherosclerosis burden) is associated with early radiographic adjacent segment degeneration after posterior lumbar interbody fusion, suggesting abdominal aortic calcification may relate to post-fusion degeneration trajectory [1]
- Abdominal aortic calcification → worse recovery after decompression for lumbar stenosis. More severe AAC was a bad sign for 2‑year outcomes [4]
- Both moderate and severe abdominal aortic calcification → reduced improvement in low back pain after surgery [5]
- Seeing abdominal aortic calcification on routine lateral lumbar X‑rays was linked with lower volumetric bone mineral density [6]
Key takeaways: Why abdominal aortic calcification matters for surgeon decision intelligence
- Abdominal aortic calcification isn’t “extra info.” It’s a risk signal about the patient’s whole-body vessel health. Abdominal aortic calcification acts like a visible clue of wider blood-vessel “wear and tear,” which can influence how well tissues tolerate stress and recover, even if the spine findings look straightforward.
- Abdominal aortic calcification helps turn outcomes into a pre-op risk profile, not a post-op surprise. Because abdominal aortic calcification can be scored from routine imaging (e.g., AAC-24 on standard lateral lumbar radiographs), it offers a practical way to capture “vascular status” that may relate to complications, delayed healing, or recovery trajectory, making it usable in real pre-op planning.
- Abdominal aortic calcification shifts planning from “what we’re fixing on the scan” to “what the body’s supply lines can handle.” Like paraspinal muscle quality reflects mechanical reserve, abdominal aortic calcification reflects the reliability of the body’s delivery system (blood flow/vascular dynamics). That framing can support more individualized decisions: approach selection, staging, optimization, and counseling based on physiologic resilience, not just anatomy.
References
[1] Sakaura H, Ikegami D, Fujimori T,Sugiura T, Mukai Y, Hosono N. Does Atherosclerosis Have Negative Impacts onEarly Adjacent Segment Degeneration After Posterior Lumbar Interbody Fusion?Global Spine J 2021;11:674–8. https://doi.org/10.1177/2192568220919370.
[2] Burkhard MD, Caffard T, Schönnagel L,Medina S, Guven AE, Mielke AM, Verna B, Chiapparelli E, Loggia G, Gregg AC, LanR, Shue J, Girardi FP, Cammisa FP, Sama AA, Hughes AP. Abdominal aorticcalcification is associated with impaired fusion after elective spinal fusion.Spine Journal 2025;25:2493–502. https://doi.org/10.1016/j.spinee.2025.04.012.
[3] Guven AE, Evangelisti G, SchönnagelL, Zhu J, Amoroso K, Chiapparelli E, Camino-Willhuber G, Tani S, Caffard T,Arzani A, Shue J, Sama AA, Cammisa FP, Girardi FP, Soffin EM, Hughes AP.Abdominal aortic calcification is an independent predictor of perioperativeblood loss in posterior spinal fusion surgery. European Spine Journal2024;33:2049–55. https://doi.org/10.1007/s00586-024-08184-y.
[4] Sakaura H, Ikegami D, Fujimori T,Sugiura T, Owaki H, Fuji T. Abdominal Aortic Calcification Is a SignificantPoor Prognostic Factor for Clinical Outcomes After Decompressive Laminotomy forLumbar Spinal Canal Stenosis. Global Spine J 2019;9:724–8. https://doi.org/10.1177/2192568219827265.
[5] Schönnagel L, Muellner M, Suwalski P,Guven AE, Camino-Willhuber G, Tani S, Caffard T, Zhu J, Haffer H, Arzani A,Chiapparelli E, Amoroso K, Shue J, Duculan R, Sama AA, Cammisa FP, Girardi FP,Mancuso CA, Hughes AP. Association of abdominal aortic calcification and lowerback pain in patients with degenerative spondylolisthesis. Pain 2024;165:376–82.https://doi.org/10.1097/j.pain.0000000000003018.
[6] Muellner M, Haffer H, Chiapparelli E,Dodo Y, Shue J, Sama AA, Cammisa FP, Girardi FP, Hughes AP. Abdominal aorticcalcification assessed on standard lateral lumbar radiographs as a screeningtool for impaired bone status in spine surgery. European Spine Journal2023;32:3030–8. https://doi.org/10.1007/s00586-023-07846-7.
