Picture two walls in the same house, built the same way, and painted the same color. Now imagine you take the same screw, and you put it into each wall in the same spot with the same force, like you’re installing a handrail.
In the first wall, the screw bites into a solid stud. It grabs tight. The rail feels steady.
In the second wall, everything looked identical… but behind the drywall the stud is quietly damaged (water-rot, tiny termite tunnels, or softened wood). You do the exact same install, but the screw doesn’t grip the same way. The rail might feel “okay” at first, but under normal everyday use it can loosen, wobble, or fail.
That’s the key idea for spine surgery:
Two vertebrae can look similar from the outside, but the “inside strength” can be very different.
Vertebral bone quality on MRI is like a special “stud finder” for the spine. It doesn’t just admire the paint; it gives surgeons a clue about what the inside of the wall is like before they place hardware – and this entirely radiation free.

What is vertebral bone quality on MRI?
Vertebral bone quality can be calculated from a standard MRI scan of the spine that makes fat appear bright (often referred to as a 'T1' scan). MRI doesn’t measure bone density directly, but it does show the signal from bone marrow inside the vertebra. When bones become osteopenic or osteoporotic, the marrow often shifts toward more fat, and that fatty marrow tends to look brighter on this MRI picture type. The MRI extracted vertebral bone quality turns that brightness into a more standardized measurement by sampling the signal inside several vertebrae (commonly L1–L4) and then normalizing it, dividing by the signal from nearby cerebrospinal fluid, so the score is less affected by differences in MRI scanners or settings. In general, a higher MRI derived vertebral bone quality score means the marrow looks relatively brighter (suggesting more fatty marrow and therefore poorer bone quality / higher osteoporosis risk), while a lower score suggests the opposite. Vertebral bone quality is useful because it can often be obtained from an MRI that a patient may already be undergoing. This helps to identify concerns about bone quality without the need for additional radiation. Furthermore, it has been studied for its potential to improve surgical risk planning for patients undergoing spinal surgery [1–6].
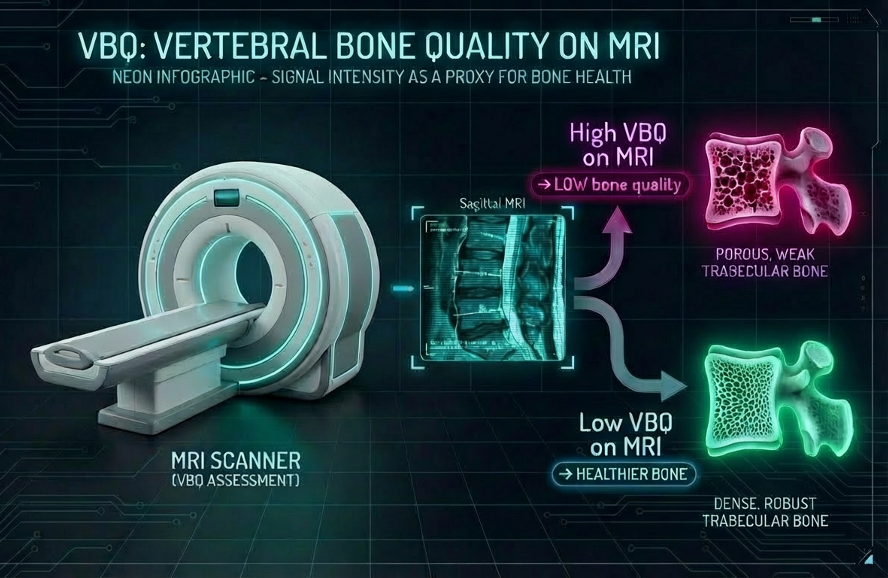
Why should we care about MRI derived vertebral bone quality?
If a surgeon is planning spine surgery, bone quality matters for example because:
- Screws need to grip the bone like a screw gripping a stud: if the bone is weak, screws may not hold as well, and surgery can be riskier
- They can adapt surgical strategy in patients with poor bone quality
- They can better anticipate complications such as screw loosening, cage subsidence, or junctional problems and planning accordingly
So, the vertebral bone quality score can help doctors do “smarter planning,” like:
- Identifying people who might have low bone density
- Deciding who might benefit from bone-strengthening steps before/after surgery
- Improving conversations about risk (what could happen, and how likely) [3]
From multiple studies we know that vertebral bone quality associates with osteopenia / osteoporosis and can help predicting surgical outcomes:
- Vertebral bone quality can help identifying osteoporosis (lumbar operative patients). Vertebral bone quality allows to differentiate healthy vs osteoporotic groups, indicating it’s use for surgical planning [1,2]
- Vertebral bone quality adds to fusion risk score. In thoracic / lumbar fusion patients, adding the vertebral bone quality score improved prediction of 90-day reoperation vs fusion risk score alone [3]
- A higher vertebral bone quality score (indicative for lower bone quality) is an independent risk factor for pedicle screw loosening following instrumented transforaminal lumbar interbody fusion [4]
- In patients undergoing adult spine deformity correction, higher vertebral bone quality score was independently associated with proximal junctional kyphosis and proximal junctional failure occurrence. Measurement of vertebral bone quality score on preoperative MRI may be a useful adjunct to adult spine deformity surgery planning [5]
- A higher cervical vertebral bone quality (C-VBQ) score is significantly associated with cage subsidence following anterior cervical discectomy and fusion (ACDF) [6]
The challenge: extracting VBQ reliably across the real world’s MRI landscape
If VBQ is so useful, why isn’t it already embedded in every preoperative MRI report? The short answer: MRI scanners are not created equal, and VBQ is a relative measurement, not an absolute one. Unlike a Hounsfield Unit on CT — which gives you a fixed number tied to a physical calibration standard — VBQ is a ratio of signal intensities that can shift depending on the scanner hardware, the software, and the imaging protocol used to acquire the scan. Different machine manufacturers, magnetic field strengths, imaging protocols, and echo times vary greatly, which “severely undermines the consistency of VBQ” across institutions [7]. Some centers use T1 FLAIR sequences (common on 3 Tesla magnets), which complicates the use of cerebrospinal fluid as a standardized internal reference signal — the very denominator of the VBQ equation.
Consider just one variable: magnet field strength. A study of 452 spine surgery patients by Lin et al. found that VBQ scores obtained on 1.5 T MRI were significantly higher than those from 3.0 T MRI — to the point that entirely different diagnostic thresholds were needed (1.5 T cutoffs of ∼3.7–3.8 versus 3.0 T cutoffs of ∼2.6) [8] . A large meta-analysis of 23 studies comprising 2,981 patients confirmed that magnet field strength is a significant source of heterogeneity in VBQ accuracy: pooled sensitivity was 0.83 on 1.5 T scanners but dropped to 0.67 on 3.0 T scanners [9]. And field strength is just one variable — add differences in coil design, vendor-specific reconstruction algorithms, and the range of reported osteoporosis thresholds across the literature spans from 2.5 all the way to 14.3 [10]. Universally standardizing imaging protocols across every hospital and outpatient MRI center is simply not feasible.
On top of the scanner variability problem, there is the human bottleneck. Calculating VBQ requires a radiologist or researcher to manually select the mid-sagittal slice, carefully place regions of interest (ROIs) within the L1–L4 vertebral bodies while avoiding cortical bone and the posterior venous plexus, and separately sample the CSF signal — a process that is time-consuming, requires specialized software, and introduces inter-rater variability [10].
This is where artificial intelligence becomes not just helpful, but arguably necessary. Recent work has shown that deep learning models can automate the entire VBQ pipeline with accuracy that rivals human experts. In one study, a YOLOv8-based AI algorithm for automated VBQ calculation demonstrated intraclass correlation coefficients (ICC) of 0.88–0.93 when compared to expert human raters — statistically indistinguishable from the human-to-human ICC of 0.95 — with Pearson correlations of 0.86–0.90 for VBQ scores [10]. Critically, AI doesn’t just replicate the manual workflow faster — it opens the door to solving the cross-scanner problem itself. Machine learning can develop automated normalization methods across scanner types and correction algorithms for field strength differences, preserving the opportunistic nature of VBQ while dramatically improving its reproducibility [7]. In other words, AI could learn to “speak the dialect” of each scanner and translate VBQ into a common language.
VBQ has strong clinical evidence behind it, but unlocking its full potential as a scalable, automated, and scanner-agnostic biomarker will likely require AI to handle both the extraction and the harmonization.
Take-home message
Vertebral bone quality is like using MRI as a “stud finder” for spine bones. Even if the outside looks okay, vertebral bone quality helps to understand whether the inside is strong enough to hold screws safely, especially useful for planning fusion surgery. Vertebral bone quality derived by MRI can add helpful information without extra radiation.
References
[1] Kim AYE, Lyons K, Sarmiento M, Lafage V, Iyer S. MRI-Based Score for Assessment of Bone Mineral Density in Operative Spine Patients. Spine (Phila Pa 1976) 2023;48:107–12. https://doi.org/10.1097/BRS.0000000000004509.
[2] Ehresman J, Pennington Z, Schilling A, Lubelski D, Ahmed AK, Cottrill E, Khan M, Sciubba DM. Novel MRI-based score for assessment of bone density in operative spine patients. The Spine Journal 2020;20:556–62. https://doi.org/10.1016/j.spinee.2019.10.018.
[3] Ramos O, Razzouk J, Beauchamp E, Mueller B, Shafa E, Mehbod AA, Cheng W, Danisa O, Carlson BC. Adding Vertebral Bone Quality to the Fusion Risk Score. Spine (Phila Pa 1976) 2024;49:916–22. https://doi.org/10.1097/BRS.0000000000004974.
[4] Hu Y-H, Chou J-H, Yeh Y-C, Hsieh M-K, Tsai T-T, Chen W-J, Chen L-H, Lai P-L, Niu C-C. The MRI-based vertebral bone quality score is a predictor of pedicle screw loosening following instrumented posterior lumbar fusion. Sci Rep 2025;15:1696. https://doi.org/10.1038/s41598-025-85625-8.
[5] Kuo CC, Soliman MAR, Aguirre AO, Ruggiero N, Kruk M, Khan A, Ghannam MM, Almeida ND, Jowdy PK, Smolar DE, Pollina J, Mullin JP. Vertebral Bone Quality Score Independently Predicts Proximal Junctional Kyphosis and/or Failure After Adult Spinal Deformity Surgery. Neurosurgery 2023;92:945–54. https://doi.org/10.1227/neu.0000000000002291.
[6] Soliman MAR, Aguirre AO, Kuo CC, Ruggiero N, Khan A, Ghannam MM, Rho K, Jowdy PK, Mullin JP, Pollina J. A Novel Cervical Vertebral Bone Quality Score Independently Predicts Cage Subsidence After Anterior Cervical Diskectomy and Fusion. Neurosurgery 2023;92:779–86. https://doi.or
[7] Kim AYE, Lyons K, Sarmiento M, Lafage V, Iyer S. MRI-Based Score for Assessment of Bone Mineral Density in Operative Spine Patients. Spine (Phila Pa 1976) 2023;48:107–12. https://doi.org/10.1097/BRS.0000000000004509.
[8] Ehresman J, Pennington Z, Schilling A, Lubelski D, Ahmed AK, Cottrill E, Khan M, Sciubba DM. Novel MRI-based score for assessment of bone density in operative spine patients. The Spine Journal 2020;20:556–62. https://doi.org/10.1016/j.spinee.2019.10.018.
[9] Ramos O, Razzouk J, Beauchamp E, Mueller B, Shafa E, Mehbod AA, Cheng W, Danisa O, Carlson BC. Adding Vertebral Bone Quality to the Fusion Risk Score. Spine (Phila Pa 1976) 2024;49:916–22. https://doi.org/10.1097/BRS.0000000000004974.
[10] Hu Y-H, Chou J-H, Yeh Y-C, Hsieh M-K, Tsai T-T, Chen W-J, Chen L-H, Lai P-L, Niu C-C. The MRI-based vertebral bone quality score is a predictor of pedicle screw loosening following instrumented posterior lumbar fusion. Sci Rep 2025;15:1696. https://doi.org/10.1038/s41598-025-85625-8.
[11] Kuo CC, Soliman MAR, Aguirre AO, Ruggiero N, Kruk M, Khan A, Ghannam MM, Almeida ND, Jowdy PK, Smolar DE, Pollina J, Mullin JP. Vertebral Bone Quality Score Independently Predicts Proximal Junctional Kyphosis and/or Failure After Adult Spinal Deformity Surgery. Neurosurgery 2023;92:945–54. https://doi.org/10.1227/neu.0000000000002291.
[12] Soliman MAR, Aguirre AO, Kuo CC, Ruggiero N, Khan A, Ghannam MM, Rho K, Jowdy PK, Mullin JP, Pollina J. A Novel Cervical Vertebral Bone Quality Score Independently Predicts Cage Subsidence After Anterior Cervical Diskectomy and Fusion. Neurosurgery 2023;92:779–86. https://doi.org/10.1227/neu.0000000000002269.