
Your Spine’s Hidden Support System: How Paraspinal Muscle Can Upgrade Surgical Decision-Making In The Future
A sailboat mast looks solid at rest, but its real test is movement and the hidden rigging that keeps it from whipping off-center. From the dock, it looks like the mast does the hard work:Standing tall, holding the sail, taking the wind. But anyone who’s sailed knows the truth: the mast only survives motion because the rigging manages it by tensioning, correcting sway, and spreading forces before a gust becomes failure.
Your spine works the same way.Vertebrae and discs are the mast, and the “rigging” is your paraspinal muscle system, built to stabilize and enable controlled movement.
So, when that rigging is impacted, and fatty infiltration rises while cross-sectional area shrinks, it’s not just a radiological detail. It can mean weaker segment-by-segment control, less physiologic load-sharing after surgery, and more mechanical stress where you least want it.
And that’s the point: if we only plan around the mast, we miss the system that determines how the spine behaves.
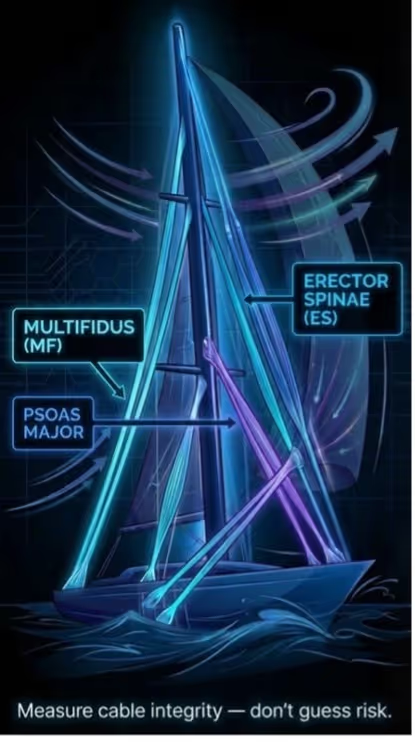
What are paraspinal muscles?
Paraspinal muscles are the muscles that run along both sides of your spine, forming a living support system that doesn’t just “hold you up,” but actively controls posture andmovement. Together, they comprise several distinct muscle groups, including deep stabilizers and more superficial extensors, that work in coordination to maintain spinal alignment and manage load. Among these, the multifidus, erector spinae, and psoas major are the key muscles highly relevant to spinal stability and function. The multifidus sits deep and close to the vertebrae, acting like a precision stabilizer that helps control small, segment-by-segment motion; the erector spinae sits more superficially and spans multiple levels, powering larger movements while also providing global support and load-sharing across the spine. The psoas major links the lumbar spine to the hip, contributing to trunk control and everyday functions like walking and lifting—so when these muscles degenerate, the spine can lose both stability and smooth, efficient motion [1].
When the “support-and-motion” system degenerates: Fatty infiltration and atrophy
Over the years, the spine’s support system changes due to the realities of life such as aging, higher BMI, and chronic strain and the paraspinal muscles can quietly shift toward more fatty infiltration and less functional muscle. Fatty infiltration is more than a cosmetic swap of “muscle for fat”: the literature notes that fatty infiltration can inhibit normal muscle activity, meaning the muscle may still be visible on MRI but contribute less to real-world control of posture and motion. At the same time, muscles can show cross sectional area shrinkage(atrophy), a true reduction in muscle size, which often reflects longer-term loss of capacity and is commonly captured through total cross-sectional area and/or “functional” cross-sectional area measures that try to isolate the lean, contractile portion of the muscle [1].
When muscle quality drops, surgical risk goes up
Once muscle quality drops, it doesn’t just describe muscle pathology, but studies show they can help predict how well patients do after spine surgery [1–7].
Worse paraspinal muscle quality is tied to worse outcomes like revision surgery risk, or fusion failure:
· > 58% fatty infiltration in the multifidus was linked to about a 2.7× higher chance of needing revision surgery for adjacent segment disease (OR 2.7, p = 0.032) [2]
· Worse function 2 years after surgery:Higher erector spinae fatty infiltration was associated with worse overall disability(Oswestry disability index: Est = 0.45, p = 0.004), and higher multifidus fatty infiltration was associated with worse standing (p = 0.033) and walking (p =0.017) [3]
· Higher chance that the decompression surgery “doesn’t hold”: 42% needed spinal fusion within 2 years after decompression, and the early-conversion group had higher posterior paraspinal fatty infiltration (53.2% vs 51.1%, p = 0.03) [4]
· Paraspinal muscle quality (muscle “quality,” not just fat/size): In 463 spine patients, higher paraspinal muscle quality- indicative for lower lean muscle quality- predicted higher pain intensity (ES OR 6.07, p = 0.033; MF OR 4.88, p = 0.019) [5]

How do we measure muscle health on MRI?
Now that we know the paraspinal muscles matter for outcomes, the next question is simple: how do we measure muscle quality in a way we can trust—and compare across patients and time?
A quantitative imaging biomarker is a number pulled from a scan (like MRI or CT) that makes “this looks degenerated” more objective, so it’s measurable, repeatable, and useful for decisions (for example: tracking change over time, comparing patients, and testing whether a measurement predicts outcomes. Good quantitative imaging biomarkers are designed to be accurate and reproducible, so the same muscle measured the same way gives a similar number each time.
From MRI to muscle metrics: The 3-step workflow
Here’s the practical part: how do we turn an MRI slice into a few simple muscle numbers
Most studies do this in a simple, repeatable way:
· Pick the slice (standard spot): Start with an axial T2-weighted MRI slice, often at the L4 upper endplate
· Draw the muscles: Outline/segment the paraspinal muscles on that slice
· Calculate the numbers of the following:
o Cross-sectional area
o Functional cross-sectional area (lean muscle)
o %fatty infiltration = fat area / total CSA
o Or the newer add-on metric paraspinal muscle quality. Instead of just measuring fat and size, the paraspinal muscle quality index uses the T2 signal of lean muscle normalized to the adjacent cerebral spinal fluid aiming to catch earlier“muscle quality” changes that might not show up yet as big fatty infiltration or functional cross-sectional area changes [5]

Key takeaways: Why paraspinal muscle quality matters for surgeon decision intelligence
1) Paraspinal muscle quantitative imaging biomarkers are not “extra info.” They are risk features that can change surgical outcomes based on underlying biological parameters.
2) Paraspinal muscle quantitative imaging biomarkers help turn outcomes into a pre-op risk profile, not a post-op surprise. Metrics like fatty infiltration, functional cross-sectional area, and newer paraspinal muscle quality signals capture tissue status that correlates with disability, pain, and revision risk, making them actionable inputs for surgeon decision-making.
3) Paraspinal muscle quantitative imaging biomarkers shift surgical planning from “what we’re fixing on the scan” to “what this patient’s support system can actually handle.” By quantifying the muscular envelope that stabilizes the spine, they help tailor the plan to the patient’s mechanical reserve, not just the bones and discs.
References
[1] Suo M, Zhang J, Sun T, Wang J, Liu X, Huang H, Li Z. The association between morphological characteristics of paraspinal muscle and spinal disorders. Ann Med 2023;55. https://doi.org/10.1080/07853890.2023.2258922.
[2] Burkhard MD, Chiapparelli E, Hambrecht J, Köhli P, Guven AE, Tsuchiya K, Schönnagel L, Caffard T, Amoroso K, Altorfer FCS, Evangelisti G, Zhu J, Shue J, Kelly MJ, Girardi FP, Cammisa FP, Sama AA, Hughes AP. Multifidus Degeneration: The Key Imaging Predictor of Adjacent Segment Disease. Global Spine J 2025;15:2348–58. https://doi.org/10.1177/21925682241300085.
[3] Hambrecht J, Köhli P, Duculan R, Lan R, Chiapparelli E, Guven AE, Evangelisti G, Burkhard MD, Tsuchiya K, Shue J, Sama AA, Cammisa FP, Girardi FP, Mancuso CA, Hughes AP. The Impact of Paraspinal Muscle Degeneration on Oswestry Disability Index Subsections Two Years After Spinal Surgery for Degenerative Lumbar Spondylolisthesis. Spine (Phila Pa 1976) 2025;50:1290–5. https://doi.org/10.1097/BRS.0000000000005192.
[4] Chiapparelli E, Caffard T, Medina SJ, Amoroso K, Zhu J, Guven AE, Evangelisti G, Hambrecht J, Kohli P, Tsuchiya K, Tripathi V, Verna B, Shue J, Sama A, Girardi FP, Cammisa FP, Hughes AP. Relationship between posterior paraspinal muscle fat infiltration and early conversion to lumbar spinal fusion following decompression surgery. European Spine Journal 2025;34:3031–7. https://doi.org/10.1007/s00586-025-09030-5.
[5] Guven AE, Finos K, Nathoo I, Köhli P, Burkhard MD, Chiapparelli E, Arzani A, Hambrecht J, Evangelisti G, Tsuchiya K, Verna B, Shue J, Sama AA, Girardi FP, Cammisa FP, Hughes AP. Introducing the Paraspinal Muscle Quality (PMQ) Score: A Novel T2 MRI-Based Intensity Parameter for Lean Muscle Assessment in Spine Patients. Spine (Phila Pa 1976) 2025. https://doi.org/10.1097/BRS.0000000000005385.
[6] Haffer H, Muellner M, Chiapparelli E, Dodo Y, Camino-Willhuber G, Zhu J, Tan ET, Pumberger M, Shue J, Sama AA, Cammisa FP, Girardi FP, Hughes AP. Georg Schmorl Prize of the German Spine Society (DWG) 2023: the influence of sarcopenia and paraspinal muscle composition on patient-reported outcomes: a prospective investigation of lumbar spinal fusion patients with 12-month follow-up. European Spine Journal 2024;33:1737–46. https://doi.org/10.1007/s00586-024-08268-9.
[7] Caffard T, Arzani A, Amoroso K, Chiapparelli E, Medina SJ, Schönnagel L, Zhu J, Verna B, Finos K, Nathoo I, Tani S, Camino-Willhuber G, Guven AE, Zadeh A, Tan ET, Carrino JA, Shue J, Dobrindt O, Zippelius T, Dalton D, Sama AA, Girardi FP, Cammisa FP, Hughes AP. Importance of the lumbar paraspinal muscles on the maintenance of global sagittal alignment after lumbar pedicle subtraction osteotomy. J Neurosurg Spine 2024;41:332–40. https://doi.org/10.3171/2024.3.SPINE231052.
